Exploring Protein Structures: Primary, Secondary, Tertiary, and Quaternary
Proteins are marvels of biological architecture, functioning as the workhorses of cells and orchestrating numerous essential processes within living organisms. The intricate functions of proteins are deeply intertwined with their three-dimensional structures. In this comprehensive article, we will journey through the hierarchy of protein structures—primary, secondary, tertiary, and quaternary—unveiling their significance and providing illuminating examples along the way.
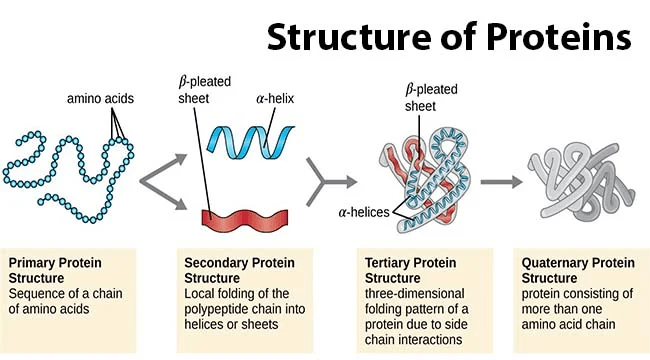
1. Primary Structure
The primary structure of a protein represents its linear sequence of amino acids, linked together through peptide bonds. This sequence dictates the unique identity and properties of each protein. A slight change in the sequence can lead to significant alterations in protein structure and function.
Example: Insulin, a hormone crucial for regulating blood sugar levels, consists of 51 amino acids arranged in a specific order.
2. Secondary Structure
The secondary structure of a protein refers to the local spatial arrangement of amino acids. It is primarily stabilized by hydrogen bonds between the peptide backbone components (amino and carbonyl groups). The two most common types of secondary structures are α-helices and β-sheets.
Example: α-helices are prevalent in proteins like keratin (a structural protein in hair and nails), while β-sheets are found in proteins like silk fibroin (responsible for the strength of spider silk).
3. Tertiary Structure
The tertiary structure is the overall three-dimensional conformation of a single protein molecule, resulting from interactions between distant amino acids. These interactions include hydrogen bonds, disulfide bonds, hydrophobic interactions, and electrostatic attractions. The tertiary structure determines the protein’s active site, where interactions with other molecules occur.
Example: Hemoglobin, the oxygen-carrying protein in red blood cells, showcases complex tertiary structure due to its multiple interacting subunits.
4. Quaternary Structure
The quaternary structure refers to the arrangement of multiple protein subunits (polypeptide chains) in a functional protein complex. This structure is relevant for proteins composed of more than one polypeptide chain. The interactions between subunits can involve the same types of forces that stabilize tertiary structures.
Example: Hemoglobin demonstrates both tertiary and quaternary structures. It is composed of four subunits—two α chains and two β chains—each with its own tertiary structure, which come together to form the quaternary hemoglobin structure.
Importance of Protein Structure
The structure of a protein is directly linked to its function. Even a slight alteration in the structure can disrupt its function. The lock-and-key analogy is often used to illustrate how proteins interact with other molecules. The protein’s structure (the lock) determines its ability to bind to specific molecules (the key) in a highly specific manner.
Moreover, the concept of denaturation highlights the sensitivity of proteins to changes in their environment, such as temperature and pH. Denaturation disrupts the intricate structure of proteins, rendering them nonfunctional.
Protein Folding and Chaperones
The process by which a protein assumes its functional three-dimensional structure is known as protein folding. It’s a remarkable feat considering the complexity of protein structures. However, protein folding isn’t always error-free. Misfolding can lead to diseases like Alzheimer’s and Parkinson’s. To mitigate this, cells employ chaperone proteins that assist in proper folding, ensuring functional and correctly structured proteins.
Conclusion
The journey through the intricate world of protein structures reveals the awe-inspiring complexity of life’s building blocks. From the linear sequence of amino acids to the intricate three-dimensional arrangements, each level of protein structure contributes to its unique functions and capabilities. Proteins are not only the engines of life’s processes but also an exquisite testament to the elegance of molecular architecture. Understanding these structures unlocks the secrets of biology, from the most fundamental cellular processes to the most intricate phenomena of life itself.



